Overview of news and achievements
Read our excitement news about finalizing work on the processing algorithms, wearable prototype development and clinical studies for the detection of cardiovascular diseases.

Seerlinq’s non-invasive technology was validated after 6 months of telemonitoring and presented in the ESC Late-Breaking Science session.

We are proud to announce that Seerlinq is now certified as a Class II(b) medical device according to the EU MDR 2017/745 regulation.

More than 200 patients, and we are collaborating with over 50 psychicians across Slovakia

Seerlinq is proud to be recognized in Deloitte Technology Fast 50 CE 2024, celebrating our innovation in healthcare!

We are still working on our research to improve the quality of our techcnology.
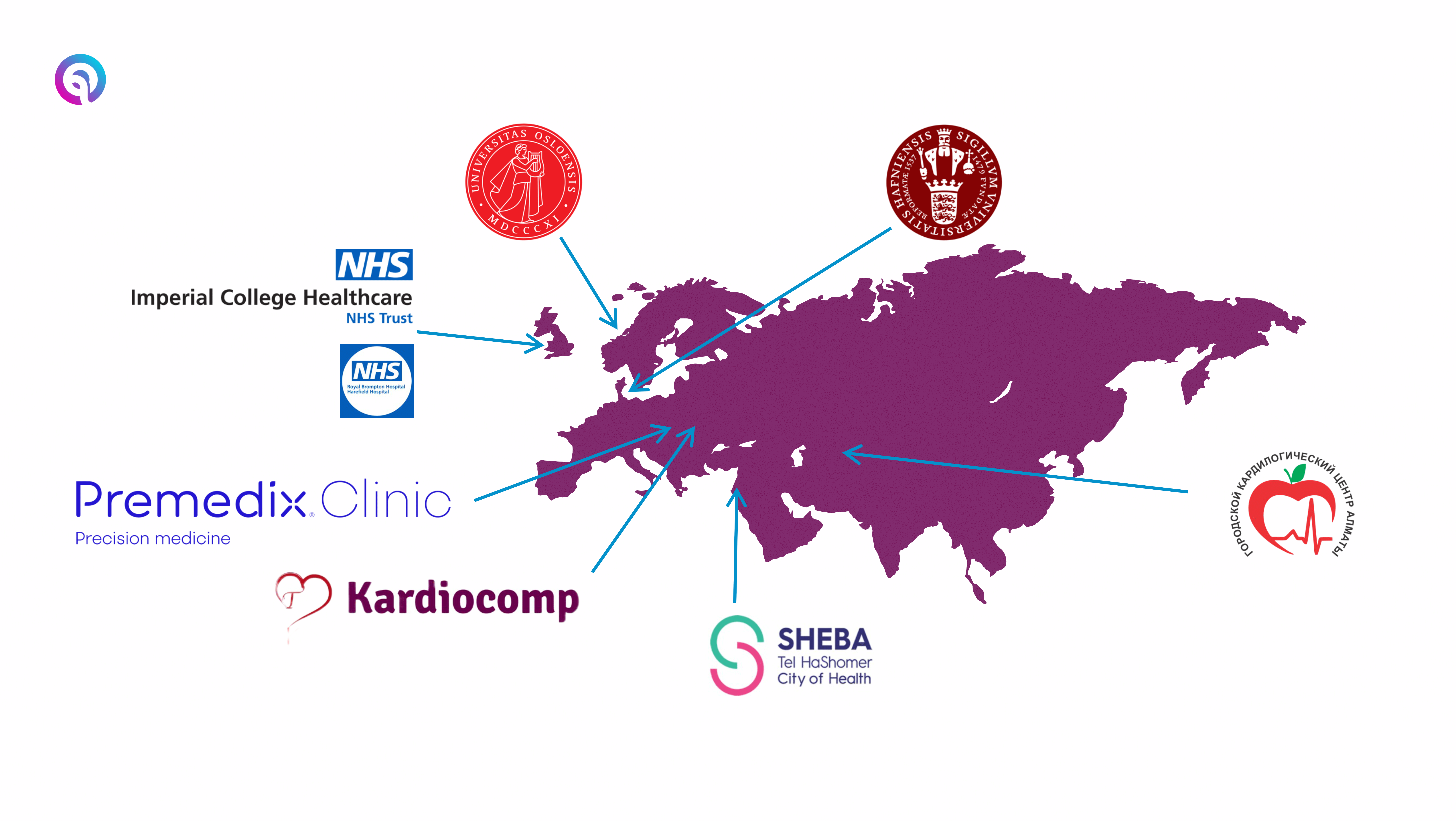
Seerlinq has an international reach!

First year for our patients without hospitalization

Improving cardiovascular care for more than 100 patients with 20 partner cardiologists

Seerlinq is now covered by all health insurance companies in Slovakia.

We’ve launched our first product for heart failure patients in Slovakia, a significant step forward in our mission!

One of the first patients successfully diagnosed and treated with our cutting-edge AI technology.

Seerlinq presented at the largest heart failure conference in the world, held in Lisbon.

Our CEO was chosen as one of the successful Slovak entrepreneurs who accompanied our esteemed President Zuzana Čaputová on a recent business trip.

Two cases were accepted for oral presentation at ESC congresses!

Clinical validation is the process that evaluates the accuracy of a medical device or a software.
Heart failure means that the heart is unable to pump blood around the body properly. It usually happens because the heart has become too weak or stiff

Photoplethysmography (PPG) is a non-invasive optical measurement method that detects changes in peripheral vascular bed.

Artificial intelligence (AI) and machine learning have the potential to revolutionize the field of medicine and healthcare.